Brain function is defined by orchestrated synaptic communication and action potential firing of neurons. Drugs (medicines) that modulate neuronal activity are used to treat a variety of therapeutic indications, including psychiatric, neurodevelopmental, neurodegenerative, epilepsy, and pain. Measuring the modulation of neuronal activity by drug candidates supports the decision-making process for drug discovery programs.
Synaptic potentials and action potentials are signals measured using electrophysiological recording techniques. At Neuroservices-Alliance, our cell electrophysiology laboratory uses three complementary recording platforms (patch clamp, microelectrode array (MEA), and fluorescence imaging) to generate functional data from neuron cultures. We use these techniques to determine the effect of small molecules, antisense oligonucleotides (ASOs), and expression of transgenes on functional neuronal endpoints (biomarkers).
We work routinely with three neuronal systems, each suited to different research questions.
- Primary rat cortical neurons (E18) are the CNS gold standard — stable, synaptically active, and well-characterized for voltage-clamp, current-clamp, and network recordings.
- Primary DRG sensory neurons, available from six or more species, are the model of choice for pain pharmacology. Species selection matters here more than is often appreciated: cynomolgus monkey and dog DRG, for example, produce dramatically different pharmacological responses to Nav1.8 blockers despite similar biophysical properties.
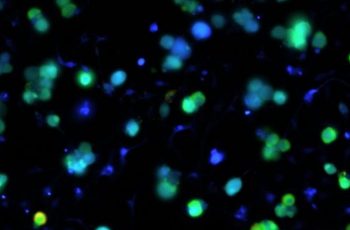
- Human iPSC-derived neurons bring direct human relevance to the assay, supporting pharmacology, AAV gene therapy validation, and maturation studies from weeks 2 through 8 in vitro.
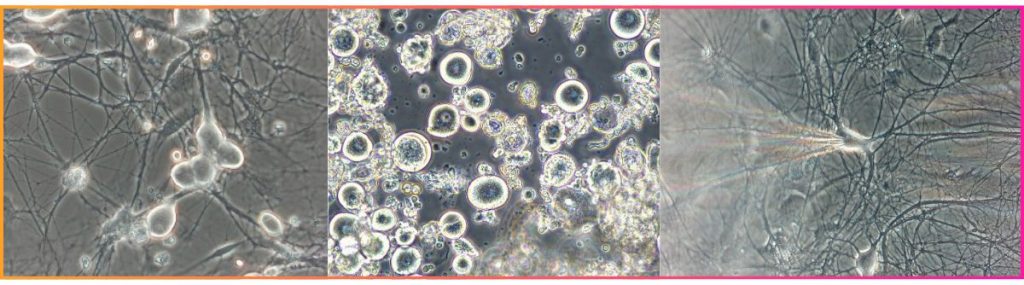
Human iPSC-derived neurons
Primary rat cortical neurons
- Fura-2 calcium imaging tracks intracellular calcium across hundreds of individually identified neurons per field of view, making it particularly powerful for TRP channel and GPCR pharmacology in sensory neurons.
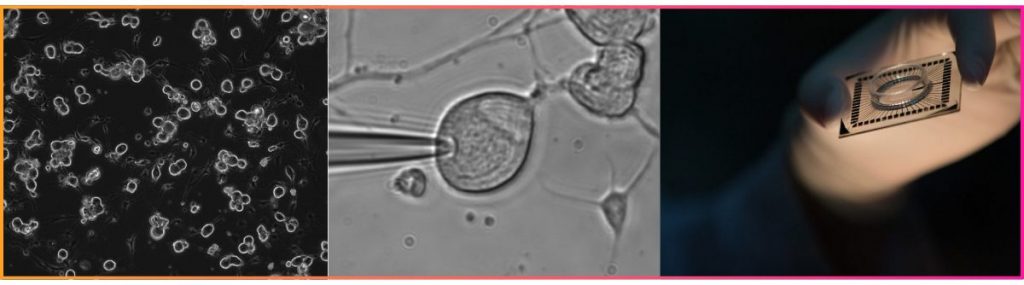
- Patch clamp offers the highest resolution, isolating individual ionic currents for IC₅₀ determination, subunit dissection, and single-cell excitability profiling.
- High-density MEA shifts the lens to the network level, recording spontaneous population activity across hundreds to thousands of electrodes simultaneously — higher throughput and ideal for CNS compound screening.
These platforms are not interchangeable — they answer different questions — but used in combination they provide a comprehensive picture of pharmacological activity across cellular and network scales.
Experimental context shapes pharmacological results in ways that are easy to underestimate. The inhibitory peptide, somatostatin, exerted a small inhibitory effect in mixed cultures containing both excitatory and inhibitory neurons. However, when the GABAergic tone was blocked with gabazine, the efficacy and potency of somatostatin was significantly enhanced. The platform matters, but so does the protocol built around it.
Cell electrophysiology at Neuroservices-Alliance is designed to be flexible enough to address early-stage screening questions and rigorous enough for translational validation.
If you’re evaluating whether our platforms are a fit for your program, our scientific team is available to discuss your specific needs and help you design the right assay for your research questions. Schedule a meeting with us today.
For a closer look at the specific assays behind these platforms, here are two posts diving into the data:
Go back