Characterizing these effects requires assays that go beyond receptor-level pharmacology and capture plasticity directly — both the structural remodeling of synapses and their functional capacity for potentiation. At Neuroservices-Alliance, we have developed a paired assay framework in our brain slice laboratory to do exactly that.

This project established two complementary plasticity assays in ketamine-treated rats, designed to capture distinct but related dimensions of synaptic remodeling.
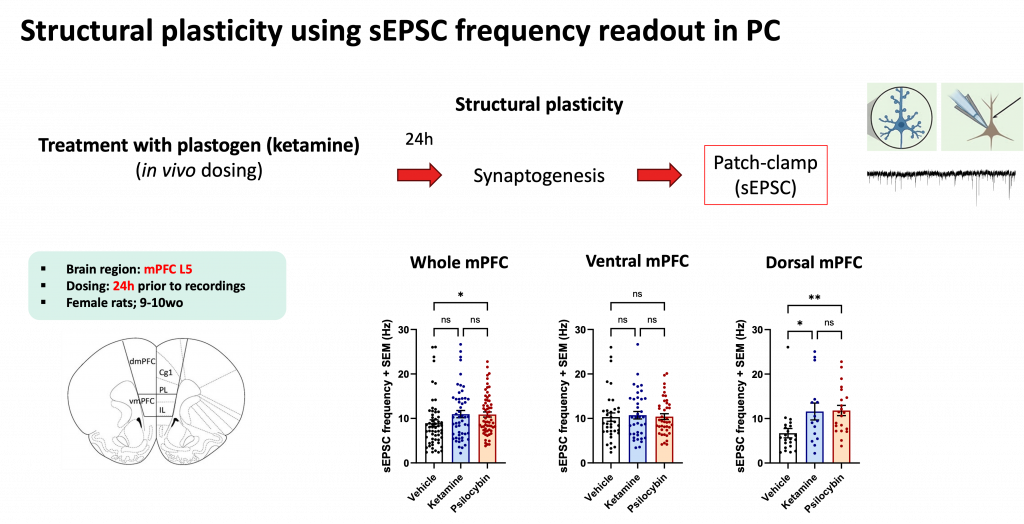
Structural plasticity is assessed through spontaneous excitatory postsynaptic current (sEPSC) frequency in whole-cell patch-clamp recordings. An increase in sEPSC frequency reflects synaptogenesis — a greater number of synaptic contacts generating more spontaneous events. Recordings were performed in layer 5 neurons of the medial prefrontal cortex (mPFC), a region central to mood regulation and a primary target of rapid-acting antidepressants.
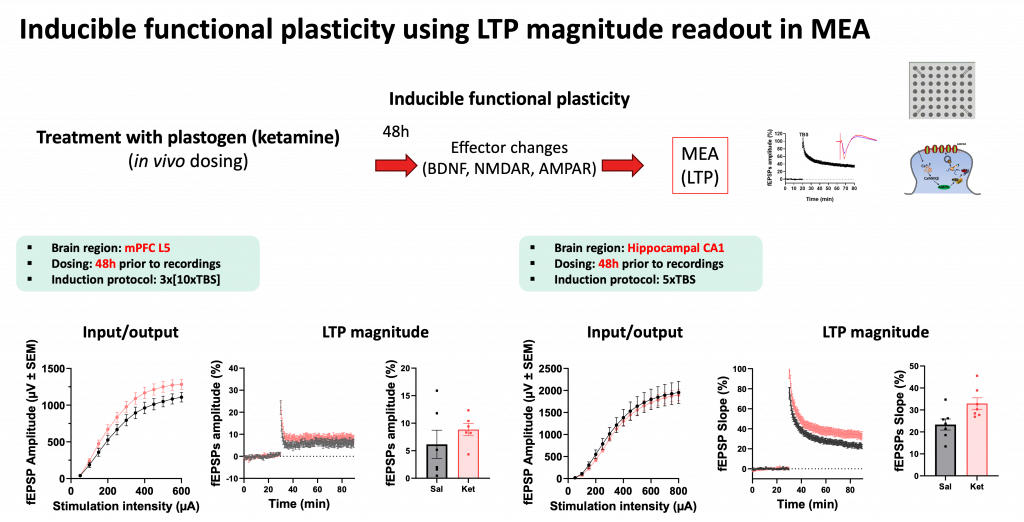
Inducible functional plasticity is assessed through long-term potentiation (LTP) magnitude using MEA slice recordings. LTP — the activity-dependent strengthening of synaptic transmission — reflects the capacity of a circuit to undergo lasting change, and is one of the key cellular mechanisms underlying learning and memory. This assay was deployed in both mPFC layer 5 and hippocampal CA1, complementing the patch-clamp data with a network-level view of plasticity.
Together, these assays provide a paired readout: structural plasticity through synaptogenesis, and functional plasticity through the capacity for LTP induction.

Female rats (9–10 weeks old) were treated in vivo with ketamine, and brain slices were prepared at defined intervals after dosing — 24 hours post-treatment for patch-clamp recordings and 48 hours for MEA recordings, consistent with the known temporal profile of ketamine’s plasticity-promoting effects.
For patch-clamp recordings, whole-cell sEPSC recordings were obtained from layer 5 mPFC neurons. sEPSC frequency was the primary endpoint, with regional analysis comparing dorsal and ventral mPFC subdivisions to capture any topographic specificity in the plasticity response.
For MEA LTP recordings, acute brain slices were placed on high-density multielectrode arrays and field excitatory postsynaptic potentials (fEPSPs) were recorded from the CA1 region and mPFC layer 5. Input/output curves were generated prior to LTP induction to confirm baseline synaptic transmission. LTP was then induced using theta burst stimulation (TBS) — 3×[10×TBS] for mPFC and 5×TBS for hippocampal CA1 — and LTP magnitude was quantified relative to pre-induction baseline. Downstream molecular correlates, including BDNF, NMDA receptor, and AMPA receptor expression, were tracked in parallel.
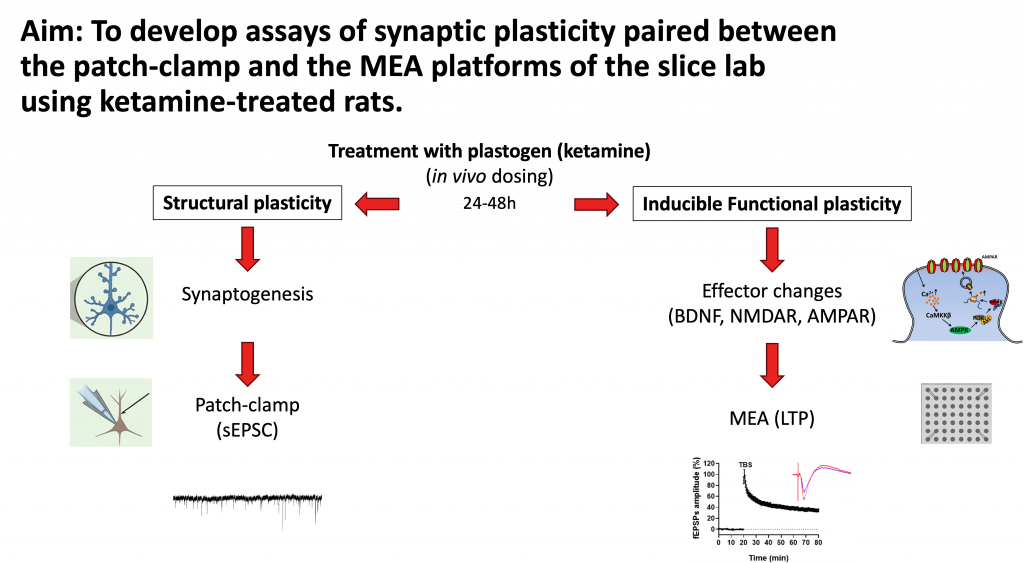

Paired patch-clamp and MEA slice assays offer a uniquely comprehensive view of synaptic plasticity — one that captures both the structural substrate and the functional output of plastogen activity. While this project used ketamine as a tool compound, the assay framework is applicable to any research program where synaptic plasticity is a relevant endpoint: antidepressant development, cognitive enhancement, neurodegeneration, or basic neuroscience.
Our brain slice electrophysiology services are designed to be flexible enough to support both drug discovery programs and fundamental research questions. Whatever the context, the science demands the same standard of rigor.
Schedule a meeting with our team to discuss how our slice platforms can support your research.
