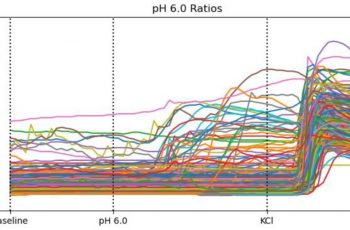

Primary rat DRG sensory neurons were analyzed using Fura-2 fluorescence calcium imaging, a well-established technique for measuring intracellular calcium dynamics in living neurons.
In this assay, neurons were loaded with the calcium-sensitive dye Fura-2, and intracellular calcium levels were monitored using the ratio of fluorescence emission at 340/380 nm excitation wavelengths. Regions of interest were selected for individual neurons, allowing calcium signals to be tracked across multiple cells simultaneously
Neurons were first recorded under baseline conditions and then exposed to pharmacological stimuli known to activate TRP channels or depolarize sensory neurons. Fluorescence ratio changes over time provided a quantitative measure of intracellular calcium responses.
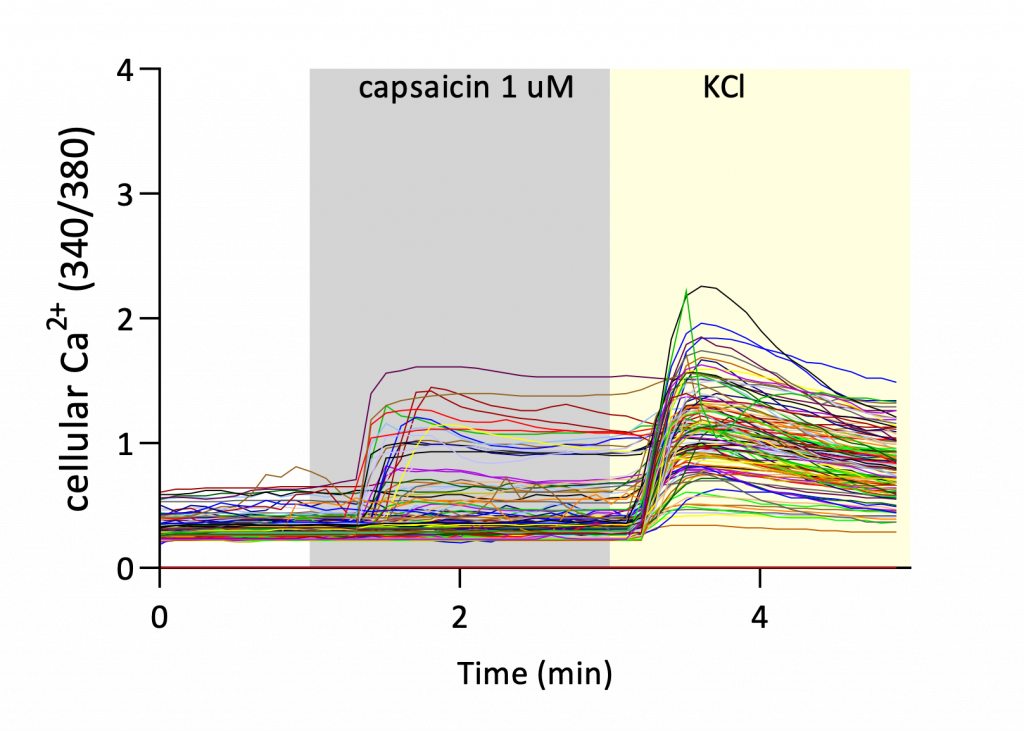
Several endpoints were used to characterize calcium mobilization and neuronal responsiveness:
Together, these endpoints allow assessment of both response magnitude and response frequency, providing a comprehensive characterization of neuronal activation
Using this calcium imaging platform, several well-characterized TRP agonists were evaluated for their ability to mobilize intracellular calcium responses in rat DRG neurons.
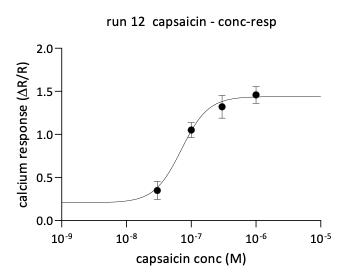
Capsaicin elicited robust calcium responses in a majority of sensory neurons. Concentration-response analysis demonstrated increasing calcium mobilization and a higher proportion of responding neurons as concentrations increased.
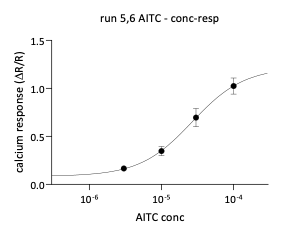
Allyl isothiocyanate (AITC) also triggered calcium responses in a subset of neurons. Increasing concentrations resulted in a higher percentage of responding neurons and increased normalized calcium signals.
Menthol stimulation elicited calcium responses in a subset of neurons, consistent with activation of TRPM8 channels expressed in sensory neurons. Concentration-dependent increases in calcium signal intensity were observed across tested concentrations.
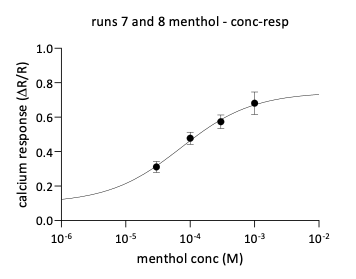
Together, these results demonstrate the ability of the assay to detect pharmacological activation of multiple TRP receptor subtypes in primary sensory neurons.
Other mediators, including histamine and bradykinin, were also tested and triggered calcium responses in subsets of neurons, demonstrating the assay’s ability to capture physiologically relevant inflammatory signaling pathways in sensory neurons.
These controls provide an important reference framework for interpreting TRP agonist responses and confirm the robustness of the neuronal culture system.

Fura-2 calcium imaging in rat DRG sensory neurons provides a powerful and quantitative platform for assessing TRP channel activation and sensory neuron signaling. By measuring intracellular calcium mobilization across neuronal populations, this assay enables detailed characterization of pharmacological responses to TRP agonists and related stimuli.